Pharmaceutical Packaging Decisions That Quietly Drive Long-Term Costs
The Short Answer: Many pharmaceutical packaging costs don’t show up on the purchase order. They show up later as labor hours, wasted materials, rework, and compliance delays that become permanent line items. The decisions you make before installation day determine whether your operation runs lean or bleeds money quietly for years.
Most packaging choices look reasonable at purchase time. But small inefficiencies compound across every unit dose packaged and every label printed, showing up as extra labor, avoidable downtime, growing scrap rates, and documentation gaps that slow audits. This guide gives you a practical way to spot those cost drivers before they become fixed expenses, whether you operate a hospital pharmacy, long-term care facility, specialty pharmacy, or contract manufacturing organization.
The Real "Cost" Is What Happens After Install Day
Upfront Price Vs. Lifetime Spend
The purchase price of pharmaceutical packaging equipment is only one piece of the total cost. What matters more is the lifetime spend, which includes labor to operate and maintain the system, consumable materials, service calls, changeover time between products, and the hours your team spends on compliance-related work. A less expensive system that requires an extra 20 minutes of setup per run or generates 5% more scrap during startup will cost far more over three to five years than a higher-priced option that avoids those losses.
Total cost of ownership in the pharmaceutical industry should account for:
- Labor per unit packaged
- Equipment uptime percentage
- Consumable waste rates
- Service and parts expenses
- Time spent on changeovers and verification
- Compliance documentation effort
Why Packaging Decisions Compound Over Time
A small inefficiency on one shift multiplies across two or three shifts, five days a week, across dozens of SKUs and multiple facilities. Add in the hours spent preparing for audits and responding to regulatory requirements, and the gap between a well-chosen system and a poorly chosen one grows wider every quarter. In the pharmaceutical sector, these compounding costs are difficult to reverse once a system is in place.
Decision 1: Unit Dose Format Choices That Affect Labor And Waste
Oral Solids Vs. Liquids, And What That Changes On The Floor
The format of your pharmaceutical product — oral solid or liquid — determines the number of touchpoints in your production process. Oral solids involve feeding, sealing, printing, and cutting individual doses, often in a blister pack or strip format where primary packaging makes direct contact with the medication. Oral liquids add filling accuracy, volume verification, and leak-proof sealing. Each additional touchpoint is a place where human error, waste, or delay can enter the workflow. The drug delivery mode also affects how the end user interacts with the finished package.
Overwrapping And Secondary Packaging
Secondary packaging, such as overwrapping or bagging, adds protection and organization. In many workflows, it reduces damage during transport and simplifies sorting at the point of drug administration. It can also support robotic dispensing. The decision comes down to matching the right packaging level to your facility’s actual needs.
What To Look For
- Number of setup steps before a run can start
- Scrap rates during the first few minutes of production
- How easy it is to isolate and correct a defect without stopping the entire line
- Whether the system handles both standard and odd-sized doses without major retooling
- Support for short-cycle dispensing models (e.g., 7-day packaging), which can reduce medication waste in long-term care settings
Decision 2: Label Design And Print Quality That Reduce Rework
Readability Is A Cost Issue
Label readability directly affects patient safety and daily efficiency. Every relabeled unit dose represents wasted material, wasted labor, and a delay in getting medication where it needs to go. In pharma packaging, where every label carries dosing, lot, and expiry information, readability failures trigger holds, manual checks, and sometimes full batch reviews.
Small Label Errors Create Big Downstream Delays
Label errors may seem minor at the point of printing, but they create outsized problems downstream. Common examples include:
- Incorrect lot or expiration date printed on a run
- Labels applied off-center or wrinkled, making barcodes unscannable
- Missing barcode or text fields due to template errors
- Wrong product label applied to the correct medication
Each of these can result in rejected product, rework, and in some cases, a quality assurance investigation. In the pharmaceutical industry, where product safety depends on accurate identification at every stage, even one mislabeled unit can trigger a broader review.
Practical Checks To Run Before You Commit
Before finalizing a labeling system or material, test under your worst-case conditions:
- Cold storage and refrigeration (does adhesive hold? does print stay legible?)
- High-humidity environments
- Abrasion from handling, transport, and automated dispensing systems
- Exposure to disinfectants and cleaning agents: common in healthcare settings
- Repeated scanning at multiple points in the supply chain
Quality packaging materials, such as thermal papers and films rated for healthcare environments, make a measurable difference in label durability and scan reliability over time.
MPI’s SUPERTHERM® thermal paper and SUPERCEL® film are built specifically for healthcare packaging environments where label durability and scan accuracy matter most.
Decision 3: Barcode And Data Workflow That Prevents Manual Workarounds
Where Costs Hide
Barcode and data problems rarely announce themselves as a single large expense. Instead, they show up as manual scanning exceptions that staff work around every day, mismatches between the data printed on primary packages and what your pharmacy or inventory system expects, and patchwork processes that differ from one site to another. These workarounds consume staff time, introduce human error, and make it harder to trace a pharmaceutical product from packaging through drug administration.
Serialization And Traceability Readiness
Even if your operation doesn’t currently require full serialization, your customers and supply chain partners may be moving in that direction. Regulatory standards around traceability continue to evolve, and many pharmaceutical company buyers now expect a unique identification code on every unit or bundle. Staying prepared means choosing packaging solutions and barcode workflows that can support these expectations without a full system replacement.
Questions To Ask Vendors
Before selecting a barcode or data workflow system, clarify the following:
- How is barcode data created, reviewed, and approved before printing?
- What happens when a barcode fails verification during a production run?
- How are corrections and changes documented for audit purposes?
- Can the system generate reports for quality assurance and regulatory compliance reviews?
MPI’s Pak-EDGE® UD Barcode Labeling Software centralizes label design, data validation, and safety checks, helping reduce the manual workarounds that drive hidden costs in barcode workflows.
Decision 4: Changeover Time And SKU Complexity That Quietly Eat Capacity
Fast Changeovers Matter More Than People Think
Every changeover between products means downtime: cleaning, resetting, loading new materials, verifying the first units off the line, and documenting the switch. For operations running dozens or hundreds of SKUs — common among pharmaceutical repackagers and specialty pharmacies — changeovers can consume a surprising share of available production hours.
Standardization Options
Look for packaging equipment that supports standardization across product formats:
- Tooling that handles multiple tablet or capsule sizes without full retooling
- Repeatable setup steps with clear, documented checkpoints
- Predefined settings for common packaging requirements that operators can load quickly
- Modular systems that scale with your needs, from R&D batches through full-scale production, without requiring entirely different equipment at each stage
Standardization reduces the risk assessment burden for each changeover and makes operator training simpler.
MPI’s Auto-Print® and Fluidose® systems are designed to handle multiple formats and batch sizes, supporting faster changeovers across oral solid and liquid packaging workflows.
A Quick Way To Estimate Impact
Here’s a simple calculation to estimate what changeovers actually cost:

Multiply by 50 weeks, and you have a rough annual cost that often surprises decision-makers. This number helps justify equipment that reduces changeover time, even if it carries a higher upfront price.
Decision 5: Uptime, Service Support, And Parts That Determine Your Real Throughput
Downtime Costs More Than The Broken Part
When a packaging line goes down, the cost extends well beyond the replacement part. Downtime triggers overtime to catch up, delayed orders, schedule reshuffles across the pharmacy or production floor, and potential quality holds on partially completed batches. In healthcare settings where medication availability directly affects patient safety, unplanned downtime has consequences that reach the bedside.
What Strong Support Looks Like
Reliable service support from pharmaceutical packaging equipment providers includes:
- Operator and maintenance training during installation and on an ongoing basis
- Remote troubleshooting to resolve issues without waiting for an on-site visit
- Parts stocked and available for fast shipment
- Documented maintenance schedules with clear intervals and procedures
- Domestically manufactured equipment and materials, which can simplify parts sourcing and reduce lead times
What To Clarify In Writing
Before purchasing, get the following in writing from your vendor:
- Response time windows for service requests
- Recommended spare parts list and pricing
- Preventive maintenance intervals
- Remote diagnostics availability and escalation process
These details are much easier to negotiate before purchase than after.
Decision 6: Compliance Documentation That Can Speed Reviews And Cut Repeat Work
Why Packaging Suppliers Matter In Compliance Planning
In the pharmaceutical industry, validation support, change control readiness, and audit trails are regulatory requirements. Equipment and materials that arrive without adequate documentation create extra work for your team. Suppliers who provide clear materials information, including details on pharmaceutical excipient contact, packaging material composition, and any materials like aluminium foil used in primary packages,help your team complete risk assessments and regulatory submissions faster.
Drug Master File Support
A Type III Drug Master File (DMF) filed with the FDA allows a packaging supplier to share confidential materials and process information directly with the agency on behalf of their customers. This means a pharmaceutical company or repackager can reference the DMF in their own submissions rather than gathering and submitting all packaging material data themselves. The result is less duplicate effort, faster review cycles, and fewer back-and-forth requests during regulatory submissions.
Decision 7: Software That Reduces Label Errors And Prevents Mismatched Data
Where Software Can Save Money
Label management software reduces cost by cutting down on manual formatting errors, redundant approval loops, and production stops from printing incorrect or outdated labels. Without centralized software controls, operations with multiple users run into version confusion and inconsistent output — both of which generate rework and waste in the pharmaceutical packaging market.
What Capabilities To Look For
When evaluating barcode labeling software for pharmaceutical packaging needs, prioritize:
- Built-in barcode rules that validate data before printing
- User permissions that limit who can create, edit, and approve labels
- Reporting tools that support audit trails and quality standards documentation
- Integration with your existing pharmacy or production systems
Pak-EDGE® UD Barcode Labeling Software
Pak-EDGE® UD Barcode Labeling Software manages barcode label design, data accuracy, and safety checks for unit dose workflows in a single platform. By centralizing label creation and applying automated validation rules, it helps reduce label errors that lead to rework, compliance findings, and manual workarounds across the packaging process.
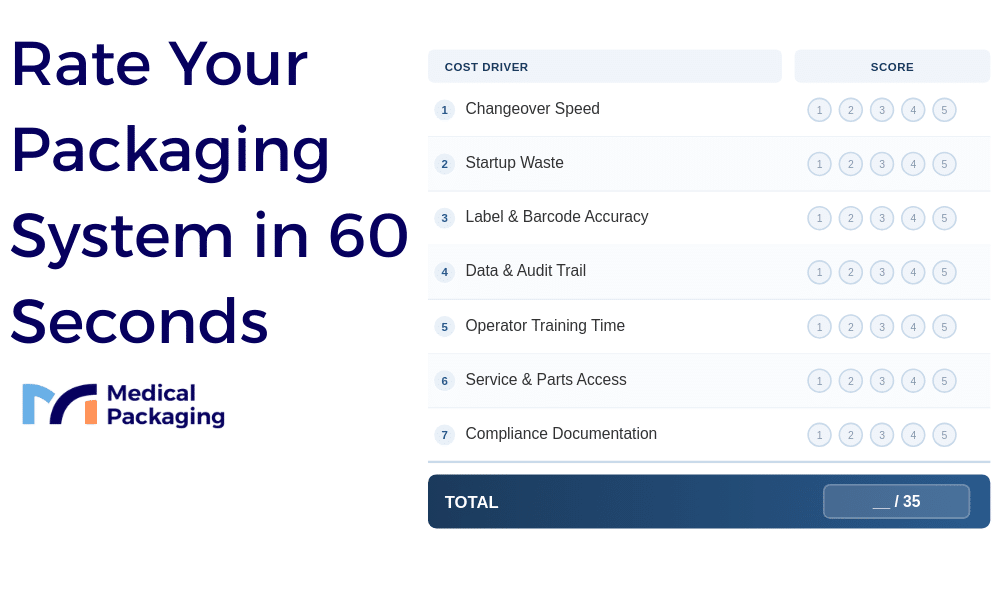
A Simple Scorecard To Compare Options
Before committing to a packaging system or supplier, score each option on a 1-to-5 scale across the cost drivers covered in this guide. This gives you a structured way to compare beyond the sticker price.
Use this scorecard during vendor evaluations, facility upgrades, or annual reviews. A low score in any single category can indicate a cost driver that will compound over time, even if the overall total looks acceptable.

Take The Next Step With MPI
Long-term pharmaceutical packaging costs come down to labor, downtime, waste, and documentation cycles. The decisions covered in this guide, from unit dose format and label quality to barcode workflows, changeover time, service support, and compliance documentation, are where those costs either grow quietly or get controlled early.
Medical Packaging Inc. LLC (MPI) manufactures unit-dose packaging and labeling systems designed to address the long-term cost drivers outlined in this guide:
- Auto-Print® oral solid packaging systems for unit dose tablet and capsule delivery
- Fluidose® oral liquid packaging systems for hospitals and specialty pharma
- PABS overwrapping for secondary packaging that supports product integrity during handling and robotic dispensing
- Auto-Wrap® and PALP labeling systems for automated syringe and accessory labeling at the point of drug administration
- Pak-EDGE® UD Barcode Labeling Software for centralized label design, validation, and safety checks
- Medical packaging materials including SUPERTHERM® thermal paper, SUPERCEL® film, and Fluidose®-specific materials
- An FDA-accepted Type III Drug Master File to support customer documentation requests and reduce duplicate regulatory effort
Run a scorecard, test your labels under real conditions, and map your data workflow end to end. Contact MPI early in the process to pressure-test your assumptions before your next packaging decision.
Content
- The Real "Cost" Is What Happens After Install Day
- Decision 1: Unit Dose Format Choices That Affect Labor And Waste
- Decision 2: Label Design And Print Quality That Reduce Rework
- Decision 3: Barcode And Data Workflow That Prevents Manual Workarounds
- Decision 4: Changeover Time And SKU Complexity That Quietly Eat Capacity
- Decision 5: Uptime, Service Support, And Parts That Determine Your Real Throughput
- Decision 6: Compliance Documentation That Can Speed Reviews And Cut Repeat Work
- Decision 7: Software That Reduces Label Errors And Prevents Mismatched Data
- Take The Next Step With MPI
Contact MPI Today for Personal Assistance
MPI’s Drug Master File provides speed-to-market regulatory and technical support related to our packaging components for medical and pharmaceutical market clients