Drug Shortages In 2026: How To Protect Patient Care When Supply Breaks
The Short Answer: Drug shortages disrupt patient care by forcing last-minute substitutions, increasing medication errors, and adding hours of manual work to pharmacy operations. The best defense is a repeatable response process that covers early detection, standardized substitutions, clean inventory, and reliable packaging and barcode controls.
Drug shortages in the United States are not new, but they remain a serious public health concern. As of late 2025, more than 200 prescription medicines were in short supply, according to the American Society of Health-System Pharmacists. While the number of new shortages has decreased from pandemic highs, the ones that persist tend to last longer and hit harder, especially generic sterile injectable drugs, cancer treatments, and other medications with limited manufacturers. For hospitals, long-term care pharmacies, and repackagers, every shortage creates a ripple effect: new products to learn, new labels to manage, new workflows to build on the fly. This guide walks through a practical, step-by-step approach to managing that fallout.
What Breaks First When A Drug Goes Short
A drug shortage rarely starts with a full “out of stock” notice. The warning signs are quieter and easier to miss.
Early Signals That Supply Is Shifting
- Backorders and allocation limits from your distributor
- Partial shipments or substituted NDC codes without advance notice
- Specific strengths or pack sizes disappearing while others remain available
- ETAs from the drug manufacturer becoming vague or repeatedly pushed back
How It Hits Patient Care
Once supply gets unreliable, the effects reach the bedside quickly:
- Med-pass timing slips as pharmacy staff spend more time sourcing and verifying unfamiliar products
- Prescriber clarifications increase when nurses encounter new packaging, different concentrations, or look-alike labels
- Dispensing delays stack up across shifts
- Manual workarounds — like hand-labeling or splitting doses — become more common
- Unfamiliar products from a new pharmaceutical company introduce look-alike/sound-alike confusion
Each of these workarounds raises the risk of medication errors. When staff are moving fast and working with products they haven’t handled before, patient safety depends on how well your systems and processes absorb the change.
Detect Shortages Early With Clear Triggers And Ownership
The earlier you spot a supply disruption, the more time you have to respond. That means defining what counts as a shortage trigger and assigning someone to watch for it.
Define Shortage Triggers
Set specific, measurable signals that tell your team a drug is at risk. Common triggers include:
- A “no ship” or “on allocation” status from your wholesaler
- Fill rates dropping below a set threshold (e.g., below 80%)
- Lead times exceeding your current on-hand window
- A noticeable rise in substitute or therapeutic alternative volume
Assign One Owner And A Backup
Shortage monitoring works best when one person owns it. Typically, procurement flags the risk, pharmacy operations confirms what’s available in-house, and a clinical lead helps set patient priority if rationing becomes necessary. Without clear ownership, early warnings get lost in email threads and shift changes.
Use A Simple Severity Scale
Not every shortage requires the same response. A three-level scale helps health care providers act proportionally:
- Level 1: Same drug, different NDC or pack size. Update item master and adjust par levels.
- Level 2: Different strength or concentration. Requires dose conversion and prescriber notification.
- Level 3: Therapeutic alternative, compounding, or rationing. Requires full clinical review and patient-level decisions.
Decide Substitutions Fast And Communicate One Standard
When a drug goes into short supply, clinical decisions need to happen quickly. But speed without structure leads to inconsistency — different shifts making different calls, outdated information circulating on nursing units, and duplicate work across departments.
Run A Small Shortage Response Group
Assign a standing group that includes pharmacy, nursing, a prescriber representative, procurement, and informatics. This team should meet weekly at baseline and shift to daily check-ins during active shortages. The goal is to make one decision and push it out everywhere at the same time.
Standardize The Decision Path
For each shortage, the group should document:
- Approved alternative(s) and the clinical rationale
- Dose conversions and any required monitoring
- Who owns follow-up if a patient was already on the affected drug
- An end date or reassessment point
This prevents individual prescribers from creating one-off substitution plans that don’t carry across shifts or units.
Publish One Source Of Truth
Medicine shortage communication matters as much as the clinical decision itself. Create a short bulletin for each active shortage that answers five questions:
- What changed?
- Who does it affect?
- What should staff do now?
- When does this take effect?
- When does it end (if known)?
Push this bulletin to order sets, dispensing systems, nursing units, and any shared reference your team uses. If the information lives in only one place or one person’s inbox, it won’t reach the people who need it at the point of care.
Keep Inventory Useful And Reduce Single-Point Failures
Shortages expose every weakness in your inventory system. Duplicate records, outdated NDC mappings, and poorly defined par levels all become bigger problems when supply is unstable.
Fix Visibility Problems First
- Clean up duplicate entries in your item master
- Correct NDC mappings and remove inactive products that cause ordering confusion
- Verify that on-hand counts reflect reality — if your system can’t show what you actually have, you’ll either over-order (tying up budget) or miss a gap until it’s too late
Protect Only The Meds That Need It
- Work with medical leadership to define a short list of medications that require safety stock
- Focus on 20–30 high-impact items where a stockout would directly delay patient care — sedation agents, vasopressors, certain antibiotics, and cancer treatments
- Adjust par levels only for that focused list; adjusting them for every drug in shortage creates chaos
Add Practical Alternatives
Reduce your exposure to any single supplier or product format:
- Qualify at least one backup supplier for high-use generics
- Identify workable alternate pack sizes that your systems can accommodate
- Evaluate alternate dosage forms (oral liquid vs. tablet, for instance) where clinically acceptable
The pharmaceutical supply chain has well-documented supply chain vulnerabilities: market concentration among a small number of drug manufacturers, reliance on overseas sources for active pharmaceutical ingredients and raw materials, and manufacturing facilities that can be disrupted by quality problems or a natural disaster. You can’t fix these upstream issues, but you can reduce how much any single failure point affects your operation.
Control Safety Risk From Packaging, Repackaging, And Barcode Changes
This is where shortages create some of their most dangerous downstream effects. Every time you switch to a new manufacturer, a new NDC, or a new pack size, your packaging and labeling environment changes. And every change is a chance for something to go wrong.
Treat Packaging And Labeling Like Safety Controls
When a new product arrives from an unfamiliar pharmaceutical company, check the basics before it reaches a patient:
- Drug name, strength, and route are clearly printed and correctly placed
- Lot number and expiration date are readable
- The label doesn’t introduce a new look-alike/sound-alike risk with other products on your shelves
Manufacturer switches during generic drug shortages are one of the most common sources of new look-alike confusion. A product that looked nothing like its neighbor on the shelf last month might now come in nearly identical packaging from a different supplier.
Build A Standard Repack And Relabel Workflow
If your facility repackages medications into unit-dose formats, shortages will increase that workload. Having a standard process prevents shortcuts that compromise traceability or patient safety:
- Intake and quarantine — Receive product, verify identity, hold until approved for repack
- Label approval — Confirm label content matches the product (drug, strength, lot, expiry, barcode)
- Verification — Scan the barcode to confirm it reads correctly before release
- Release with documentation — Record lot and expiry at every handoff, with clear hold/release steps
Identify your “high-frequency shortage meds,” the products that consistently go in and out of short supply and always generate repack work. Having templates and label formats ready for those drugs saves hours of setup every time a new round hits.
Keep Barcode Scanning Working
Barcode failures at the point of care defeat the purpose of scanning technology. Common causes include:
- Poor print contrast or smeared/wrinkled labels
- Incorrect data encoded in the barcode
- Barcode placement that scanners can’t reach easily
Set your barcode rules once — symbology, encoded data, and template change control — then log scan exceptions and fix root causes. A spike in failures after a product switch is a clear signal the new label needs attention.
Documentation And Templates That Keep Up With Constant Change
Shortages generate a repeating cycle of paperwork: substitution rationale, label revisions, staff training notes, and validation records. Without templates, teams rewrite the same documents from scratch every time a new shortage hits — wasting time and increasing the chance of missing a step.
Build Three Reusable Templates
- One-page shortage summary — Drug affected, severity level, approved alternative, effective date, and communication plan
- One-page substitution approval — Clinical rationale, dose conversion, monitoring requirements, and responsible pharmacist sign-off
- One-page repack batch record checklist — Product identity, label verification, barcode scan confirmation, lot/expiry documentation, and release authorization
Keep Traceability Tight
Every handoff in the shortage response process should capture lot and expiration data. If a product needs to be held or recalled, you need to trace it backward through every step — from the patient to the shelf to the supplier. Clear hold/release steps in your documentation prevent partially verified products from reaching the floor.
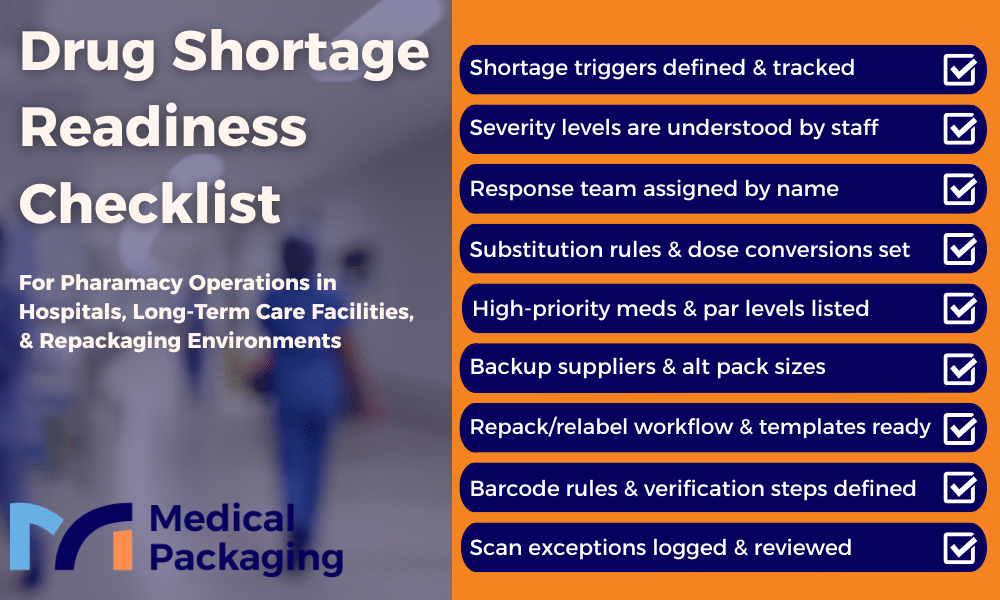
Shortage Readiness Checklist
Use this checklist to measure your current state. Any unchecked item is a gap worth closing before the next shortage hits.
How MPI Helps Teams Stay Ready
Medical Packaging Inc. LLC (MPI) builds the packaging, labeling, and software systems that support pharmacy teams through product changes and supply instability:
- Auto-Print® Oral Solid Packaging Systems — Package and barcode tablets and capsules into unit dose formats efficiently, reducing manual handling during high-volume repack situations
- Fluidose® Oral Liquid Packaging Systems — Fill and label oral liquid unit doses with accuracy and consistency, serving hospitals and specialty pharma operations
- PABS Overwrapping Systems — Provide secondary packaging for medications, supporting storage, handling, and distribution requirements
- Auto-Wrap® Syringe Labeling Systems — Automate syringe and accessory labeling for consistent, readable identification across product changes
- Pak-EDGE® UD Barcode Labeling Software — Design and manage barcode labels for medication packaging, supporting barcode control when products and NDCs change frequently
- FDA-Accepted Type III Drug Master File — Supports documentation tied to packaging components, helping clients meet regulatory review requirements and move products to market faster
Build The System Now So Shortages Hurt Less Later
Drug shortages are a supply chain problem, but the fallout lands on pharmacy staff, nurses, and patients. Every shortage adds labor, increases the risk of medication errors, and strains systems that were built for stable supply — not constant change.
A repeatable process reduces that fallout.
- Detect supply disruptions early.
- Substitute consistently using a single decision path.
- Keep inventory data clean so you can trust what your system tells you.
- Control packaging, labeling, and barcode reliability so that every product that reaches the bedside scans correctly and looks right.
The health care system will continue to face drug shortages driven by market concentration, low prices for generic drugs, quality issues at manufacturing facilities, and dependence on overseas raw materials and active pharmaceutical ingredients. These are industry-level problems that require policy solutions, increased domestic manufacturing investment, and coordination between the public and private sectors.
What you can control is how your facility responds. Run the Shortage Readiness checklist above against your current workflow, close the biggest gaps first, and contact MPI to discuss how the right packaging and labeling systems can support your shortage readiness.
Content
- What Breaks First When A Drug Goes Short
- Detect Shortages Early With Clear Triggers And Ownership
- Decide Substitutions Fast And Communicate One Standard
- Keep Inventory Useful And Reduce Single-Point Failures
- Control Safety Risk From Packaging, Repackaging, And Barcode Changes
- Documentation And Templates That Keep Up With Constant Change
- Shortage Readiness Checklist
- How MPI Helps Teams Stay Ready
- Build The System Now So Shortages Hurt Less Later
Contact MPI Today for Personal Assistance
MPI’s Drug Master File provides speed-to-market regulatory and technical support related to our packaging components for medical and pharmaceutical market clients