The Pharmaceutical Packaging Market and the Rise of Ready-to-Administer Oral Liquids

The global pharmaceutical packaging market is undergoing a significant transformation. As the pharmaceutical industry adapts to evolving patient demographics, stricter regulatory requirements, and rising demand for safer medication delivery, one trend stands out: the rapid growth of ready-to-administer (RTA) oral liquid unit dose formats.
For hospitals, long-term care pharmacies, specialty pharmaceutical manufacturers, and CDMOs, this shift represents both a challenge and an opportunity to advance patient safety and operational efficiency. Medical Packaging Inc., LLC (MPI), a leading global manufacturer of unit-dose pharmaceutical packaging and labeling systems, delivers innovative packaging solutions that help healthcare providers meet the demands of this changing market.
The Evolving Global Pharmaceutical Packaging Market
The global pharmaceutical packaging market size continues to expand, driven by increasing demand for pharmaceutical products that prioritize accuracy, compliance, and patient-centered delivery. Key factors accelerating growth across the pharmaceutical sector include:
- Aging populations in the United States, Europe, and the Asia Pacific region requiring more complex medication regimens, often involving liquid formulations for patients who cannot swallow oral solid doses
- Expanding clinical need across pediatric care, behavioral health, and geriatric medicine for pre-measured oral liquid formats that reduce dosing variability at the point of care
- Tightening regulatory standards from regulatory bodies, including the FDA, CMS, and USP, requiring greater traceability, barcoded labeling, and documentation throughout the supply chain
The primary packaging segment, the materials and systems in direct contact with pharmaceutical products, remains the primary segment driving technological advancement across the industry. Major players in the global market and specialized manufacturers like MPI each address distinct segments of this expanding pharmaceutical packaging market.
MPI focuses specifically on unit dose packaging and labeling systems for oral solids, oral liquids, and syringes, serving healthcare markets that require precision automation and regulatory compliance at every step.

What Are Ready-to-Administer Oral Liquids?
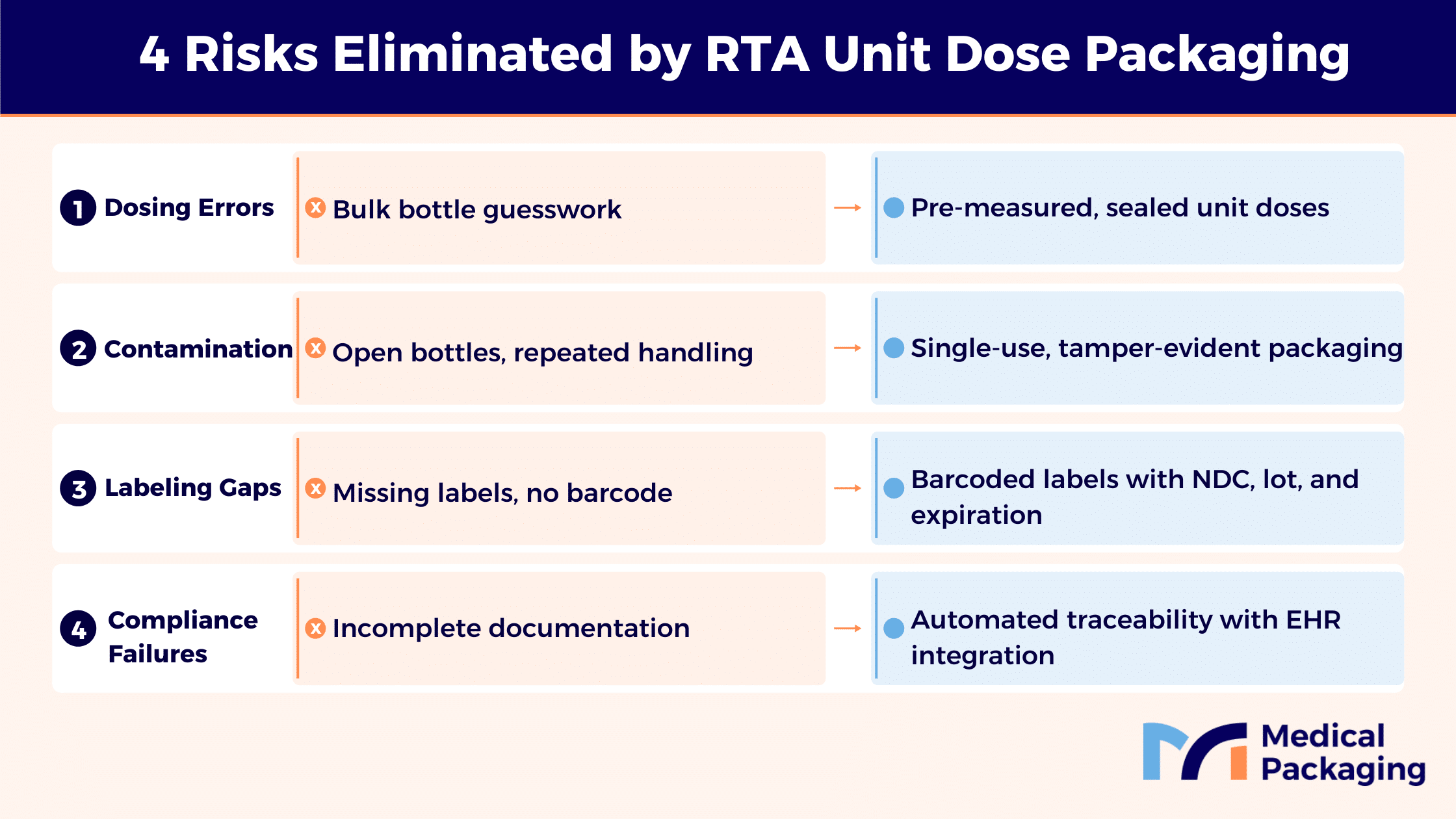
Ready-to-administer oral liquids are pre-measured, pre-labeled unit dose medications packaged in formats that eliminate manual preparation at the bedside. Rather than requiring nursing staff to measure liquid from bulk plastic bottles, a process prone to dosing errors and contamination risk, RTA formats deliver the exact prescribed volume in a sealed, barcoded package ready for immediate administration.
Key benefits of RTA oral liquid formats:
- Removes measurement variability and reduces medication errors
- Enables barcode medication administration (BCMA) scanning at the point of care
- Streamlines compliance documentation for healthcare providers operating under strict regulations from the FDA and CMS
- Simplifies audit readiness across hospital and long-term care settings
The rising demand for RTA formats reflects a broader shift across every pharmaceutical company and healthcare facility: moving from manual, error-prone processes toward automated, standardized systems that protect patients and reduce liability.
How Advanced Packaging Systems Supports the RTA Movement
Transitioning to RTA oral liquid formats requires packaging equipment capable of precise fill volumes, integrated labeling, and consistent output across varying production scales. MPI’s oral liquid packaging systems are engineered for these exact requirements.
Precision Oral Liquid Filling and Labeling
MPI’s Fluidose® systems fill and label oral liquid unit doses and syringes with accuracy validated for hospital and specialty pharma applications.
Barcoded Labeling and Regulatory Compliance
MPI’s Pak-EDGE® UD Barcode Labeling Software provides full control over label design, data entry, and barcode generation for unit dose packages. Core capabilities include:
- Comprehensive reporting for regulatory compliance
- Machine-readable data on each label: medication name, dosage, lot number, expiration date, and NDC
- Easy-To-Use, Comprehensive Label Designer
- Built-In Pre- and Post-Packaging Verification Systems
- Enhanced Security Features
- Built-In Drug Serialization and Lot Tracking
This level of traceability satisfies regulatory requirements from the FDA, CMS, and USP while reducing the risk of labeling errors that compromise patient safety.
Scalable Solutions Across Healthcare Settings
MPI’s systems offer flexibility across diverse healthcare markets:
- Hospitals and acute care facilities leverage high-throughput Fluidose® equipment for volume production
- Extended care and long-term care pharmacies manage complex multi-patient regimens with individualized unit dose packaging
- Specialty pharmaceutical manufacturers and CDMOs rely on MPI’s equipment for consistent, quality-controlled packaging of custom formulations
MPI’s FDA-accepted Type III Drug Master File further supports clients in accelerating product time-to-market and achieving regulatory compliance.

Advancing Medication Packaging with MPI
The rise of ready-to-administer oral liquids reflects a fundamental shift in how the pharmaceutical industry approaches medication safety, efficiency, and compliance. For hospitals, long-term care pharmacies, specialty pharma manufacturers, and CDMOs, adopting RTA formats supported by advanced unit dose packaging systems is essential to meeting the standards set by regulatory bodies and the expectations of healthcare providers.
MPI’s comprehensive range of solutions includes:
- Fluidose® oral liquid filling system
- Pak-EDGE® UD Barcode Labeling Software
- FDA-accepted Type III Drug Master File support
Contact Medical Packaging Inc., LLC today to explore how MPI’s oral liquid packaging and labeling systems can strengthen your medication safety program and support your operational goals.
Contact MPI Today for Personal Assistance
MPI’s Drug Master File provides speed-to-market regulatory and technical support related to our packaging components for medical and pharmaceutical market clients