Navigating the Complexities of Clinical Trial Logistics & Packaging
Clinical Trial Logistics at a Glance: Clinical trial logistics covers the movement, packaging, storage, and timely delivery of investigational medicinal products to investigator sites worldwide. For pharmaceutical companies, trial sponsors, and clinical research teams, getting logistics right means protecting patient safety, meeting regulatory requirements, and keeping studies on schedule.
Clinical trial logistics is the backbone of every successful clinical study. Packaging plays a direct role in these outcomes, as the way clinical trial materials are packaged, labeled, and tracked affects regulatory compliance, product integrity during transit, and accuracy at the point of administration. That’s why pharmaceutical packaging is not a downstream consideration in trial logistics, but a factor that influences planning from the very beginning.
The National Institutes of Health (NIH) defines a clinical trial as a research study in which human subjects are assigned to interventions, whether medications, surgery, medical devices, or behavioral therapy, to evaluate effects on health-related outcomes. Each type of intervention brings its own packaging, labeling, and distribution demands.
Who Is Involved in Clinical Trials?
Running a clinical study requires an experienced clinical research team that typically includes doctors, nurses, social workers, and other healthcare professionals filling roles such as principal investigator, research pharmacist, and study coordinator. In global clinical trials, the team extends to logistics providers and regional coordinators responsible for moving investigational medicinal products and ancillary supplies to investigator sites across multiple countries. Packaging specialists also play an important role, as the materials used to package and label medicinal products must meet the protocol requirements of each trial and the regulatory requirements of each region.
Sponsors, often referred to as the trial sponsor, keep science moving forward by providing funding. These sponsors may include:
- Pharmaceutical company investments in research and development, with the industry spending over $83 billion in 2019 according to the Congressional Budget Office
- Academic medical centers, health maintenance organizations, and other healthcare institutions
- Voluntary organizations, individuals, and companies
- Federal agencies such as NIH, the U.S. Department of Defense, the National Science Foundation, and the U.S. Department of Health and Human Services
Where and at What Scale Do Clinical Trials Take Place?
Clinical trials can take place in hospitals, universities, healthcare offices, and community clinics. In the United States, investigator sites range from large academic medical centers to smaller independent research facilities. It is common for trials to run at multiple locations to recruit a diverse participant pool, and as trial logistics grow more complex, selecting the right sites and establishing reliable distribution services and packaging workflows for each one becomes a significant operational consideration.
Researchers have traditionally performed clinical trials in North America, Western Europe, and Oceania. Scientists are increasingly performing global clinical trials in other regions, expanding the global network to improve participant diversity while demanding greater global expertise in logistics, packaging, and regulatory compliance across borders.
Clinical trials vary widely in scale:
- Regional studies held in a small geographic area with a limited number of investigator sites
- National studies, like those performed by the National Institutes of Health in the U.S., requiring coordination of clinical supply across a single country
- Global studies with large, diverse participant pools that require coordination among multiple service providers and logistics providers across different regulatory jurisdictions, each with their own packaging and labeling standards
The Drug Development Process
Discovery & Development
Researchers discover new drugs through a variety of paths, from gaining new insight into rare diseases to leveraging breakthroughs in gene therapy and biologics. These advances have led to innovative medicinal products that work in new ways, such as manipulating genetic material or delivering medical products to targeted sites within the body.
Thousands of compounds are potential candidates at this stage, but only a handful move into development. The development phase involves testing to determine optimal dosage, route of administration, side effects, drug interactions, effectiveness compared with similar drugs, and effects across different population groups.
Clinical Manufacturing
Clinical manufacturing, also known as clinical trial material manufacturing (CTM), focuses on producing pharmaceuticals for use in clinical trials. This stage is a significant portion of clinical trial supply services, as each pharmaceutical company must manage production to meet both protocol requirements and regulatory requirements.
Manufacturing for clinical trials differs from standard production in two important ways: volume is typically much lower, and flexibility must be much higher. Formulation changes and regulatory updates can shift plans quickly, and trial materials may need to be repackaged, relabeled, or reformatted as the clinical study evolves. Having adaptable packaging systems matters from the start.
While the FDA does not require a finished drug product in clinical trial material, the federal agency does recommend using disposable medical equipment, pre-sterilized containers, closed process equipment, and current good manufacturing processes (cGMP) facilities and testing laboratories.
Supply Chain Management in Clinical Trials
The clinical trial supply chain covers drugs, materials, medical equipment, ancillary supplies, packaging, and storage. Understanding and optimizing each link helps confirm that trials can run without interruption.
The clinical logistics market reflects the growing importance of this work. According to Grand View Research, the global clinical trial supply and logistics market was valued at approximately USD 4.29 billion in 2024 and is projected to reach USD 6.72 billion by 2030, growing at a CAGR of 7.97%. This growth is driven by tightening regulations, rising demand for logistic solutions, and the increasing complexity of clinical supply needs, from cold chain storage and transportation to packaging and distribution services. Cardiovascular disease remains one of the leading therapeutic areas driving clinical trial activity, alongside oncology and rare diseases.

Logistical Challenges in Clinical Studies
Running a clinical trial presents unique challenges, even for experienced teams. Supply chain disruptions, highlighted by the COVID-19 pandemic, have underscored the need for resilient trial logistics and agile distribution services. Availability of raw materials, transportation delays, and allocation of medical supplies can all slow or stop clinical trials.
Cost
As biotechnology and biopharmaceutical manufacturing continue to evolve, they are becoming the focus of global clinical trials across therapeutic areas such as cardiovascular disease, oncology, and rare diseases. The cost of supplies, medical equipment, logistic services, regulatory compliance, and complex procedures all increase when dealing with biologics and advanced therapies like gene therapy. Building a cost-effective clinical trial supply chain means incorporating trial supplies early, packaging and labeling products correctly with dosage and expiry date, and establishing local and international supply pipelines. Each pharmaceutical company and trial sponsor must weigh these costs against timely delivery and adherence to the highest standards of quality control.
Environmental Control
Biopharmaceutical products, biological samples, and gene therapy products tend to be highly sensitive to temperature ranges and can become easily contaminated. Maintaining cold chain integrity throughout storage and transportation is a major logistical challenge, and one that is difficult to address without the right packaging solutions. Sensitive shipments of investigational medicinal products often require real-time temperature monitoring and specialized handling from pickup to final delivery.
Regulatory Requirements
Regulations govern study protocol development, participant selection, informed consent, adverse event monitoring, and results reporting. These regulatory requirements protect human subjects and promote the scientific validity of clinical trials. In the United States and across international markets, regulatory compliance is mandatory for the success of clinical operations, and trial sponsors must meet both local and international protocol requirements at every stage.
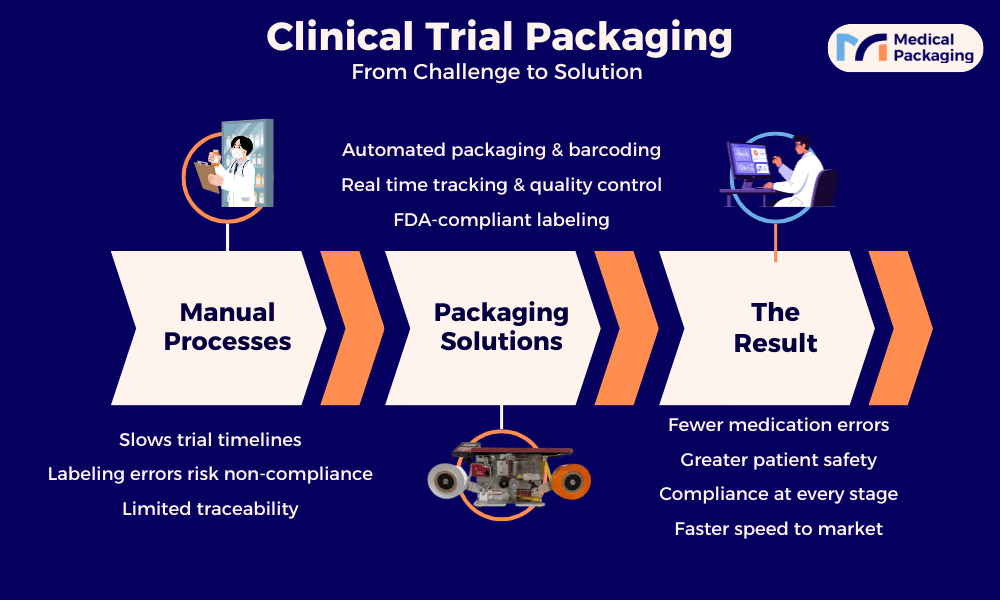
The Role of Packaging in Clinical Trial Logistics
Packaging and materials are indispensable components of the clinical trial supply chain, directly supporting the safe delivery of investigational medicinal products (IMPs) to investigator sites. Clinical trial materials increasingly involve biologic drugs administered via injection, which carry more stringent packaging requirements. A growing number of trials also use direct-to-patient distribution services, which changes packaging needs and makes real time tracking even more important.
Today’s clinical trial packaging must:
- Protect product quality during storage and sensitive shipments
- Support quality control and traceability from production through delivery
- Meet the labeling and barcode requirements of each market and regulatory body
- Adapt to changes in formulation, dosage, or protocol requirements mid-trial
Creating a Resilient Clinical Trial Supply Chain
Following industry best practices, a resilient clinical trial supply chain should incorporate:
- Diversity – working with several suppliers, service providers, and transportation affiliates to reduce single points of failure
- Efficiency – delivering quality, cost-effective products with timely delivery across all investigator sites
- Compliance – confirming that all suppliers and logistics providers meet regulatory requirements to support the highest standards of patient safety
- Traceability – tracking the movement of clinical supply and drugs in real time for transparency and confidence in your trial
How Medical Packaging Inc., LLC Serves the Clinical Trial Market
Medical Packaging Inc., LLC (MPI) is a valuable partner in clinical trial logistics and clinical operations. With medication packaging solutions tailored to the unique challenges of clinical trial supply services, MPI systems promote the highest standards of efficiency, regulatory compliance, and traceability throughout manufacturing, transportation, and administration.
As a leading manufacturer of unit dose packaging and labeling systems, equipment, software, and materials, our logistic solutions help deliver the right dose to the right patient at the right time. They also minimize possible contamination during transportation and handling of sensitive shipments, while decreasing the probability of medication errors and protecting costly medicinal products.
FD-Pharma® Unit Dose Packaging System
MPI’s FD-Pharma® features a built-in printing and pumping system, creating an automated barcoding packaging solution for unit-dose oral liquid medication. With a small footprint and competitive price point, this system offers precise dosing at speeds of up to 35 cups per minute. FD-Pharma® is appropriate for clinical trials of all sizes, from low production to full production modes, all in a single machine. The optional die cut feature trims the lidding to the perimeter of the cup, leaving one tabbed section for easy opening.
Stakeholders throughout the clinical trial process, from the trial sponsor to investigator sites, appreciate the FD-Pharma® for its ability to reduce packaging costs and decrease medication errors. Read more about the features, options, and benefits of our FD-Pharma® system.
Fluidose® Series 6 Unit Dose Packaging System
The MPI Fluidose® Series 6 is an automated barcoding packaging solution for unit-dose oral liquid medication with a built-in syringe pump mechanism. Designed to adhere to strict technical guidelines, this system produces up to 22 doses per minute. MPI-certified materials confirm that contents are protected from environmental effects such as light or moisture during storage and transit, with a clear, legible, fully-sealed unit dose package produced every time to support quality control from production through delivery.
Packaging That Provides Speed-to-Market Capabilities
In drug development and testing, delays cost time and money. MPI’s Type III Drug Master File (DMF) allows us to avoid typical delays and enables us to rapidly support customers’ regulatory compliance and filing needs, as well as meet the needs of pharmaceutical drug manufacturers and contract drug manufacturing organizations (CDMOs) for FDA-compliant liquid cup packaging.
Clinical Trial Packaging Solutions Built for Speed, Accuracy, and Compliance
When it comes to clinical trial logistics and the packaging materials that support clinical research, Medical Packaging Inc. is a trusted partner for pharmaceutical companies, CDMOs, and trial sponsors across the industry. Whether you need logistic solutions for a small-scale clinical study or full-production packaging for global clinical trials, MPI has the products and global expertise to support your work. For more information about clinical trial supply, trial logistics, and packaging materials, contact MPI.
Content
- Who Is Involved in Clinical Trials?
- Where and at What Scale Do Clinical Trials Take Place?
- The Drug Development Process
- Supply Chain Management in Clinical Trials
- Logistical Challenges in Clinical Studies
- Creating a Resilient Clinical Trial Supply Chain
- How Medical Packaging Inc., LLC Serves the Clinical Trial Market
- Packaging That Provides Speed-to-Market Capabilities
- Clinical Trial Packaging Solutions Built for Speed, Accuracy, and Compliance
Contact MPI Today for Personal Assistance
MPI’s Drug Master File provides speed-to-market regulatory and technical support related to our packaging components for medical and pharmaceutical market clients